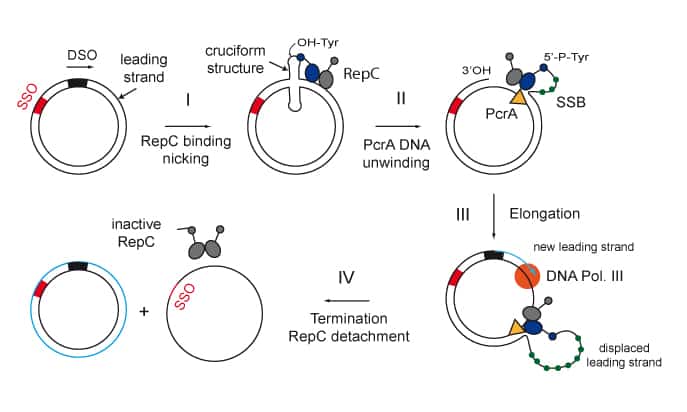
DNA breaks are highly toxic lesions, and failure to repair them correctly is associated with genomic instability leading to cell death, cancer and developmental defects. Therefore, cells possess robust mechanisms to repair DNA breaks. One such DNA repair mechanism is homologous recombination where the sister chromatid is used as a template for faithful repair. Recombinational repair is initiated by DSB resection to form a long 3´-terminated ssDNA overhang. During the last years we have focused on the bacterial model system of Bacillus subtilis and in particular in the long-range resection reaction catalyzed by the AddAB helicase-nuclease. We now aim to employ all this acquired experience and knowledge to tackle the challenging problem of understanding DNA-end processing for repair by following the real-time processing of DNA by human repair proteins using novel single-molecule approaches.
How chromosomes replicate, segregate and faithfully transmit genetic information remains a big question in contemporary cell biology. In this context, unravelling the mechanisms of DNA cohesion and condensation is a particularly challenging task because these processes are fundamentally mechanical in nature and thus particularly difficult to monitor using conventional biochemistry. Moreover, bulk biochemical analyses of an unsynchronized population of DNA/protein molecules cannot assess intermediate conformational states, cannot provide quantitative information on intermolecular forces and cannot easily discriminate between the many disparate models proposed for chromosomes organisation. A major goal of my group is to understand chromosome organisation in bacteria.
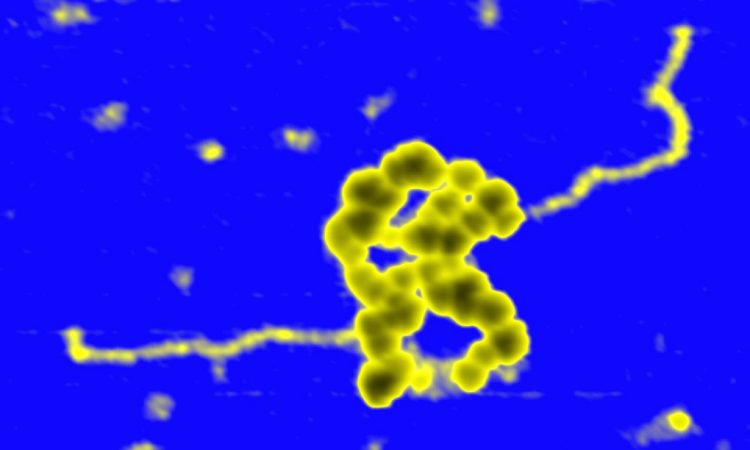
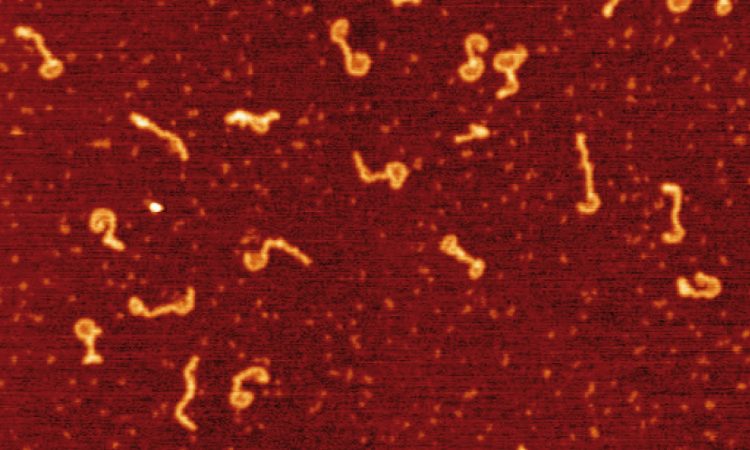
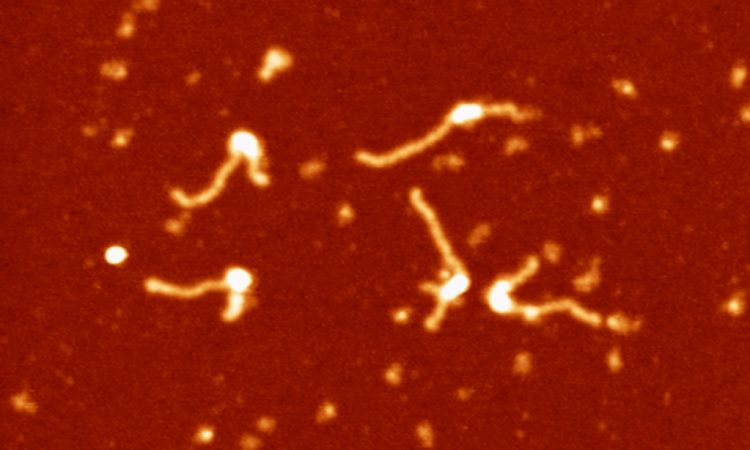
During the last years, we have built a series of single molecule setups that have provided us with the tools to investigate multiple DNA-protein interactions and mechanics of nucleic acids. Single-molecule strategies that combine high-throughput Magnetic Tweezers with fluorescence are perfectly tailored to study DNA-end processing and DNA condensation and cohesion in contrast with conventional biochemistry. Moreover, fast AFM technologies may complement the MT data with very useful structural information. Single-molecule manipulation and imaging techniques allow direct observation of protein:DNA intermediates, potentially in real time, and the study of individual enzymatic cycles in an unsynchronized population. A major goal of my group is to push to the state-of-the-art our single molecule techniques based on Magnetic Tweezers by incorporating multichannel flow platforms, epifluorescence and TIRF microscopy, and to advance our AFM setups by incorporating new technologies for fast imaging and acquisition.