Bacillus subtilis MutS modulates RecA-mediated DNA strand exchange between divergent DNA sequences
Begoña Carrasco†, Ester Serrano†, Alejandro Martín-González, Fernando Moreno-Herrero and Juan C. Alonso.
Natural transformation contributes to acquisition and spread of antibiotic resistance and pathogenicity traits. We show that Bacillus subtilis chromosomal transformation decreased logarithmically with increased sequence divergence up to 15% in wild type (wt) cells or in cells lacking MutS2 or mismatch repair proteins (MutL, MutS or both).
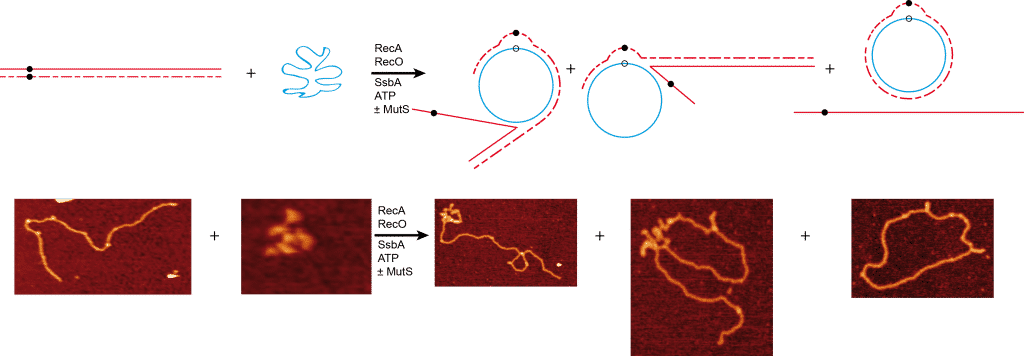
Beyond 15% sequence divergence, the chromosomal transformation efficiency is ~100-fold higher in ΔmutS and ΔmutSL than in ΔmutS2 or wt cells. In the first phase of the biphasic curve (up to 15% sequence divergence), RecA-catalyzed DNA strand exchange contributes to the delineation of species, and in the second phase, homology-facilitated legitimate recombination might facilitate the restoration of inactivated genes. To understand how MutSL modulates the integration process, we monitored DNA strand exchange reactions using a circular single-stranded DNA and a linear double-stranded DNA substrate with an internal 77-bp region with ~16% or ~54% sequence divergence in an otherwise homologous substrate. The former substrate delayed, whereas the latter halted RecA-mediated strand exchange. Interestingly, MutS addition overcame the heterologous barrier. We propose that MutS assists DNA strand exchange by facilitating RecA disassembly, and indirectly re-engagement with the homologous 5’-end of the linear duplex. Our data supports the idea that MutS modulates bidirectional RecA-mediated integration of divergent sequences, which is important for speciation without compromising genetic diversity.
